This year, as the worlds of science, technology and literature marked the bicentenary of Mary Shelley’s Frankenstein, in some laboratories human cells were being coaxed into a tiny version of the organ that most defines us.
Depending on where you sit, these "minibrains" – formally known as cerebral organoids – could rival Shelley’s creation on the monstrosity scale, raising deep moral questions about consciousness and the nature of humanity.
But they also promise hope of a cure for illnesses ranging from childhood epilepsy to Alzheimer’s disease and brain cancer.
I’ve come to Melbourne’s Florey Institute of Neuroscience and Mental Health to hunt a minibrain down. I’m expecting to find it in a dish, on a bench or perhaps in a fridge.
As it happens, the first one we stumble upon is in the bin.
A special bin for biowaste, of course. The neural tissue has died after serving its purpose and is now merely a smudge of pink on a plastic slide.
Still, it seems inconceivable that this miracle of science could qualify as rubbish.
“Some of these kids … have 20 seizures a day”: Florey Institute director Steven Petrou.Credit:Pat Scala
The contrast becomes more extreme when I sit down to chat with Florey Institute director Professor Steven Petrou. He is leading research that creates organoids to mimic the behaviour of the brains of children with rare, debilitating forms of epilepsy.
“Some of these kids can’t speak, are not mobile, they sit in a cot, they have 20 seizures a day and they die when they’re 12. So, absolutely devastating neuro-developmental disorders,” he says.
The researchers take skin cells from the children, turn them into pluripotent stem cells that can form almost any tissue in the body, then direct them to become neurons.
Through a microscope you can clearly see the slender bodies of those brain cells afloat in a watery matrix. Hook them up to electrodes and you get something mind-bending. These guys are talking to each other – the computer shows spikes of electrical activity as the neurons fire.
Steven Petrou gives a lecture in May on his work with minibrains and the Genetic Epilepsy Team Australia.Credit:Facebook
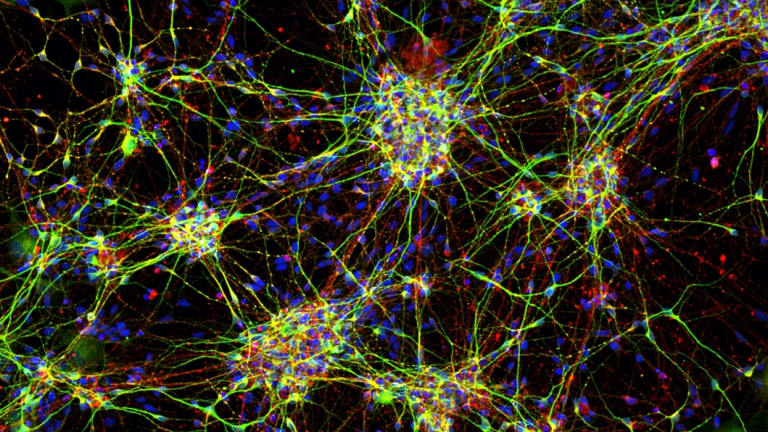
But for the kids Petrou is trying to help, the chatter is out of whack. Some have a mutation in a gene called SCN2A that controls the passage of sodium in and out of the neuron.
“This is a gain of function of excitation, so this channel works too hard and produces epilepsy,” says Petrou.
Replicating that glitch in a dish has allowed the researchers to tailor a treatment right there on the bench; Petrou is on the verge of announcing a clinical trial of a gene therapy to treat one variant of the disorder.
And it won’t just aim to stop the seizures.
“The idea with precision medicine in this application is if you can fix the fundamental disorder far enough back in the pathological chain, you should fix all the problems," says Petrou.
If the treatment works, these kids could be spared the intellectual disability and movement disorders that go hand-in-glove with constant seizures.
From Down syndrome to Alzheimer's disease
Organoid research is also making its mark at the other end of life.
Professor Ernst Wolvetang at the University of Queensland (UQ) is making brain organoids with skin cells from people with Down syndrome who, tragically, have a much higher risk of Alzheimer’s disease.
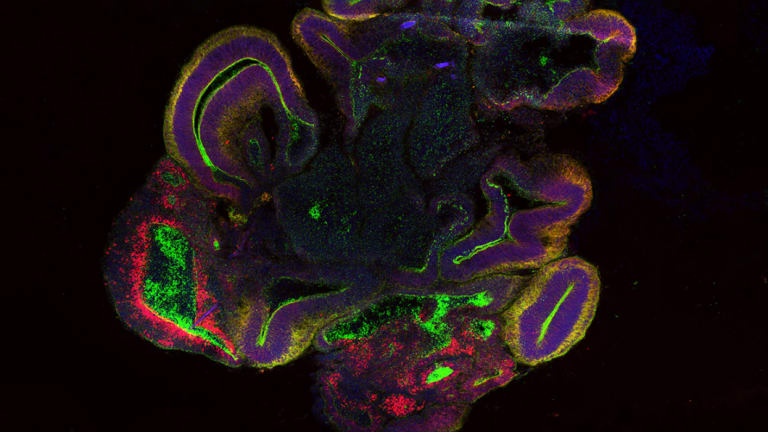
A 34-day-old brain organoid section.Credit:Courtesy of Mariana Oksdath Mansilla, Centre for Cancer Biology, Adelaide
But that also means their organoids are an exceptional model to examine the illness, which is the most common form of dementia.
“I’m still amazed and surprised by how many cell types we can find in these still relatively primitive organoids. All the major cell types that you expect in the brain are there,” says Wolvetang.
That faithful rendering has led to a major discovery.
Traditional thinking is that smudge-like amyloid plaques in the brains of people with Alzheimer’s cause a second change called tau tangles, which look a bit like musical crotchet notes. The tau tangles kill the brain cells. Wolvetang’s team used gene editing to stop plaques forming in the organoids.
What they found next has upended the Alzheimer’s orthodoxy.
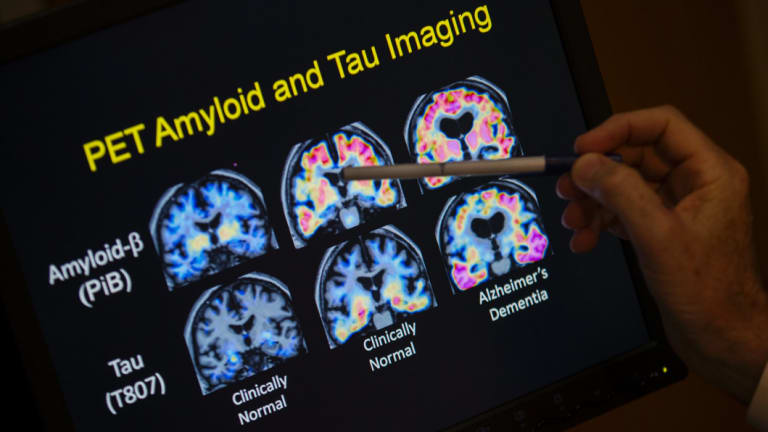
PET scan results from a study on brain function in Alzheimer’s disease at Georgetown University Hospital in Washington. Credit:AP
“The plaques are normalised but the tau pathology is still there suggesting that … the plaques are not driving that pathology directly. Which is kind of heretical but, you know, that’s how it is,” says Wolvetang, with a chuckle.
The upshot is that Wolvetang is going after tau, testing a drug in the organoids to stop it in its tracks. So does the drug, which he can’t name for commercial reasons, do the trick?
“The initial data suggest that it does,” says Wolvetang.
A light in a rat's eye
The next step is to test it in mice, but to understand how Wolvetang will do that you need a brief lesson in some recent, slightly disturbing science.
Wolvetang’s organoids grow to the size of a small pea – around five millimetres – in about 160 days. After that they begin to rot from the inside out. It’s because the minibrain has to suck nutrients and oxygen from the fluid in the dish which can only penetrate so far.
That’s why the race is on to get blood vessels into organoids.
In April, a team led by Fred "Rusty" Gage at the Salk Institute announced they had done just that. They grafted a human brain organoid into a mouse brain. The mouse blood vessels penetrated the organoid and the nerve cells of each intertwined and relayed messages.
It sounds creepy, but the ick factor ratchets up when you consider related research published in the last northern winter in the US.
Boris Karloff, left, and Colin Clive in a scene from the 1931 classic film Frankenstein. Credit:Universal Studios/AP
Scientists implanted human brain organoids into the visual centre of a rat brain. When they shone a light in the rat’s eye, the human brain cells fired. Mary Shelley would be impressed.
Horror scenarios notwithstanding, Wolvetang is following suit, grafting his organoids into mice to test, among others, if the tau drug is best given by mouth or nose and to see if it crosses the blood-brain barrier.
But there might be another way to get vessels into minibrains, one that could free the organoid to grow bigger than a mouse’s skull, essential if we’re ever to have a full-size brain on a bench.
There is a push to grow artificial blood vessels in organoids, and Wolvetang has skin in that game. Together with UQ bioengineer Justin Cooper-White, he is using primitive endothelial cells to urge vessels forth into the organoid. So, are vessels invading the minibrain?
“It is not super-ready yet, but it seems to be going that way, yes,” says Wolvetang.
Which raises the question of just how big it can get.
“I think it would be able to be at least two to three times bigger,” says Wolvetang. “It is scalable, so we don’t know exactly where the limit is.”
It is a game where size matters. As Cambridge-based organoid pioneer Dr Madeline Lancaster explained in a recent talk, novel medical treatments often fall off the “clinical trial cliff” – they might work in mice, but when you trial them in people they either don’t work or have toxic side-effects.
Stem cell-derived neurons from a four-year-old child with a mutation in the SCN2A gene causing epileptic encephalopathy. Credit:Courtesy of Florey Institute of Neuroscience and Mental Health
Hence the need for a faithful model that will approach the magnitude of the roughly 86 billion neurons in our own noggins.
But it is also about structure, and here the cerebral organoid is Janus-faced. On the one hand, it can spectacularly mimic human brain anatomy, such as the six layers of the cortex, the regions responsible for thought, speech and movement.
On the other hand its architecture can be like a Salvador Dali dreamscape, with six or seven hemispheres, two cerebellums and three retinas. It is a structure, says Lancaster, much like the childhood nerve cell tumour neuroblastoma.
That fact lends a certain irony to the work of Dr Guillermo Gomez at the Centre for Cancer Biology in Adelaide, who is deploying organoids to combat the devastating brain cancer glioblastoma.
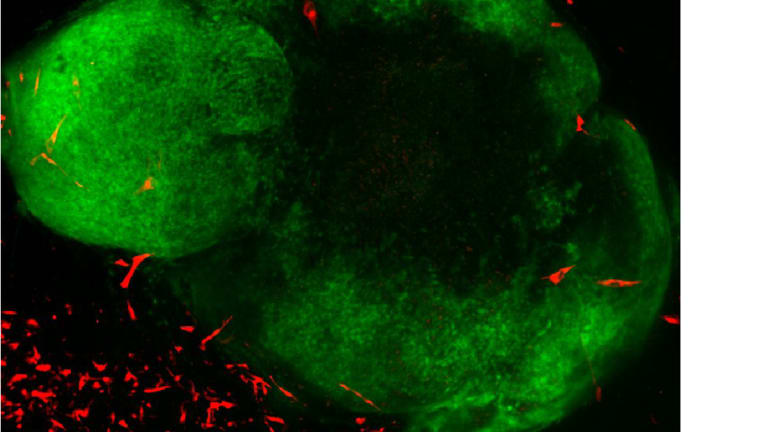
An image showing GBM (Glioblastoma multiforme) tumour cells (red) in a healthy brain organoid (green).Credit:Courtesy of Mariana Oksdath Mansilla, Centre for Cancer Biology, Adelaide
“There is no cure,” says Gomez of the cancer, whose five-year survival rate is 4.6 per cent.
“With the five-year survival rate during the last 30 years, you see a flat curve. No changes. No progress,” he says.
Gomez is aiming to shift that trend. He takes samples of glioblastoma, removed when the patient has surgery for the tumour, then regrows the cancer in a brain organoid made from healthy stem cells.
The goal is to test drugs that might halt the tumour’s spread while leaving the healthy brain tissue intact. It is precision medicine writ large.
“Each person is different … diseases are also very different. It is very hard to put everyone in the same bag. That is why we cannot give the same drug to every patient,” says Gomez.
“The research starts in the patient and is aimed to be used in the same patient.”
'Our organoids are dreaming'
The hope of stunning medical breakthroughs aside, there is something unsettling about the fact that when these scientists go home at night, the organoids sit quietly in their wells firing away.
It’s a point that hasn’t escaped Wolvetang.
“Our organoids are dreaming. Of course they are not dreaming like we do, but they have what we call autogenic activity. They are firing by themselves, which makes you wonder why they are talking to each other,” he says.
As that chatter gets more complex we will, according to US neuroscientist Christof Koch, be faced with a very thorny question: “Is this thing in pain?”
It is the kind of question UNSW philosopher Professor Susan Dodds ponders for a living. Her response in this case is, however, sceptical.
“Even fully developed adult brain tissue does not itself feel pain,” says Dodds.
She is referring to the glaring fact that, once neurosurgeons have cut through the skull, they can operate on the brain while a person is wide awake because it doesn’t have pain receptors.
Affinity, a massive, interactive light sculpture representing neurons in the brain is seen on the forecourt of the Arts Centre in Melbourne in 2015. The sculpture was commissioned by Alzheimer’s Australia. Credit:Wayne Taylor
Of course, direct electrical stimulation of the brain in the right spot can trigger sensations, including pleasure. And, although pain is complex, researchers have recently identified an “ouch zone”. So, could a zap to the right spot in the organoid cause pain?
“When you experience pain, you require a brain that is able to process it as suffering,” says Dodds.
“The big question is do the brain organoids have consciousness, and I don’t think we have any evidence of consciousness whatsoever with these things.”
What we do have, says Dodds, are patterns of activity that we might compare with the wriggling of a roundworm, well short of anything that could qualify as a conscious self.
She also points to another reason why a sentient organoid is a very big ask.
“In order for it to have sentience it needs to be integrated with some kind of external experience … we would normally think that sentience requires some kind of organ of sense,” she says.
And organoids, of course, don’t come equipped with ears, eyes, nose, tongue or curious exploring fingers. Yet.
The question of consciousness
An experiment posted on preprint website bioRxiv in June shows just how fast the field is moving. A team led by Lancaster connected a human brain organoid to a mouse spinal cord with attached muscles.
By stimulating the organoid they created an impulse that made the muscle contract. It’s hard not to think of Dr Frankenstein yelling “It’s alive” as his monster’s fingers twitch in the 1931 horror movie.
The point being that with an outgoing motor pathway established, the possibility for an incoming sensory pathway would seem to be closer. I asked Lancaster about that via email.
“I do think it might be feasible to provide sensory input of some sort,” says Lancaster, with some caveats.
“Sentience is much more than just input and output,” she says, adding that you need complex neuron types, chemical messengers such as dopamine, and an organised flow of information over the network.
“A cockroach has all of the above, but we wouldn’t consider it to have advanced cognition … and it’s generally accepted that in addition to sentience our advanced cognition is what makes us human,” says Lancaster.
Stem cells shown through a microscope in a research lab at the Gladstone Institute of Cardiovascular Disease in San Francisco in 2010. Credit:Bloomberg
Adding to the imbroglio, even if a minibrain could get conscious, how would you ever know? After all, you can’t ask it if it is awake.
As it happens, a technique is being refined to objectively measure consciousness. In a recent book, Italian neuroscientist Marcello Massimini describes a procedure, which he first published in 2013, that stimulates the brain from outside the skull and measures the brainwave response.
Massimini has calibrated it on people with varying degrees of impaired consciousness, from sleep to severe brain trauma. As consciousness drops, brainwaves become less complex.
Last year he modified the technique to work with ferret brain slices kept alive in a solution (clearly the human calibrations don’t apply). I asked him via email whether that technique could, in principle, work in organoids.
“Yes, it could be used,” said Massimini, adding that “[T]he aim is clearly to explore the mechanisms of complexity rather than inferring consciousness.”
Complexity is something of a buzzword in organoid research. Recent Australian research upped the stakes by 3D printing brain tissue, bringing a replica brain on a bench into tantalising – if very distant – focus.
But Petrou stresses just how tricky that task is going to be.
“We know how fragile a real brain is. One genetic mutation, some trauma, and that brain doesn’t work anymore,” he says. “It is so easy to break and therefore that means it is probably going to be so difficult to reproduce.”
Nonetheless, as the technology advances you wonder if those organoids could grow to the point where one day they start to gain bona fide moral status.
If that happens, the first sign might just be a very slight pang as scientists toss their dreaming minibrains into the garbage.
Source: Read Full Article