Even though neurodegenerative diseases often strike in middle age or later, patients could have structural differences in their brains that arise before birth.
In a new study in Cell Reports, USC stem cell scientists used both patient-derived nerve cells and laboratory mice to demonstrate that the most common genetic cause of amyotrophic lateral sclerosis, (ALS) and frontotemporal dementia (FTD) impairs neural stem cells and reduces the size of two key regions of the brain during embryonic development.
“We are only beginning to understand how neurodevelopment in an embryo can contribute to neurodegeneration in an adult,” said the study’s corresponding author Justin Ichida, who is the John Douglas French Alzheimer’s Foundation Associate Professor of Stem Cell Biology and Regenerative Medicine at the Keck School of Medicine of USC, and a New York Stem Cell Foundation-Robertson Investigator. “Our study offers some suggestive evidence of how this might occur in the context of ALS and FTD.”
Up to 10% of cases of ALS and FTD result from mutations in a gene called C9ORF72, even though the two diseases usually manifest very differently. Whereas ALS, also known as Lou Gehrig’s disease, causes progressive paralysis and death within three to five years of diagnosis, FTD most commonly affects behavior, personality and language.
Both diseases are usually diagnosed in middle age or later, and yet carriers of the C9ORF72 mutation can show reductions in the size of two brain regions—the thalamic region and the frontal cortical region—decades before developing any symptoms.
“It’s unclear whether this reduction in gray matter arises from altered development in an embryo or from early degeneration in an adult,” said first author Eric Hendricks, a postdoc in the Ichida Lab.
Evidence of altered development in the embryo
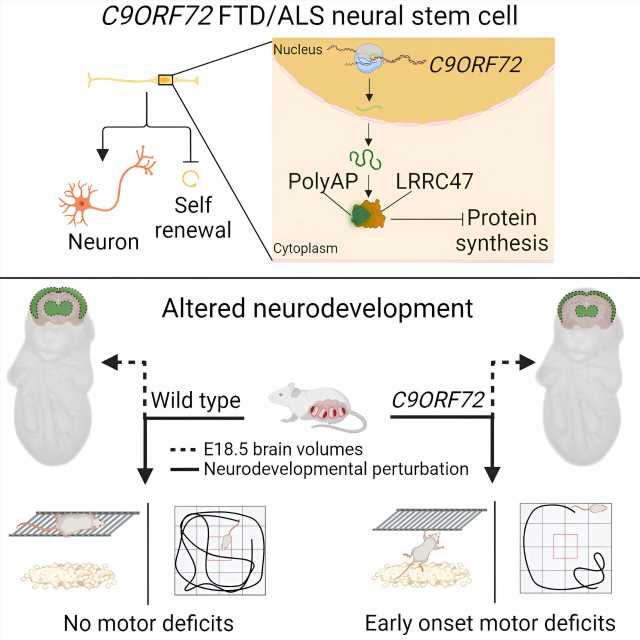
Hendricks and his colleagues approached this question by taking skin or blood cells from patients with ALS or FTD resulting from mutations in C9ORF72, and reprogramming these cells into neural stem cells, which are the cells that form the nervous system during embryonic development.
Compared to neural stem cells derived from healthy people, the neural stem cells from patients with ALS or FTD weren’t able to renew their population. Instead, the patient-derived neural stem cells had a tendency to prematurely differentiate into mature neurons.
The patient-derived neural stem cells contained a mutant protein called poly(AP). Although poly(AP) isn’t fatally toxic to cells, it does impair their ability to make proteins necessary for building new cells. As a result, poly(AP) impairs neural stem cells’ ability to renew their population.
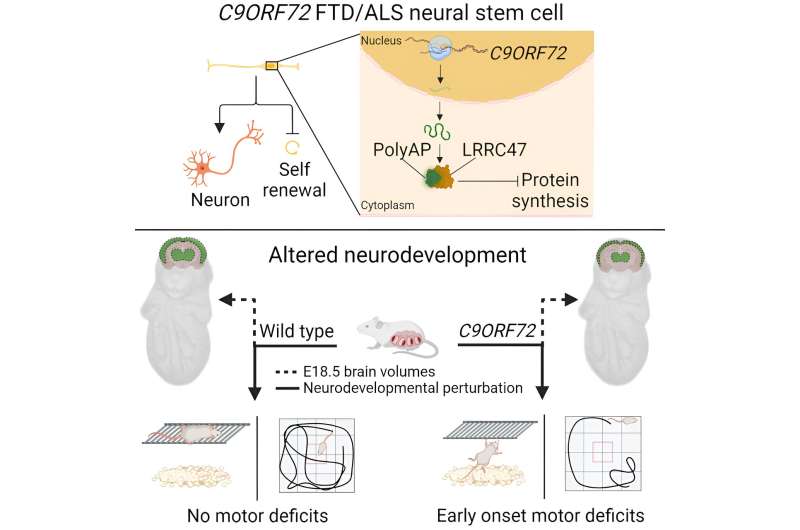
The scientists wanted to understand the consequences of a non-renewing population of neural stem cells in a living organism, so they turned to laboratory mice.
In embryonic mice, mutations in C9ORF72 caused measurable developmental changes not only in their brains, but also elsewhere in their bodies. In their brains, these changes included smaller thalamic regions, as well as reduced cortical thickness. The mice also weighed 5–10% less at embryonic day 18.5.
In addition, if scientists administered a drug that caused similar structural changes to the brain during embryonic development, mice experienced clumsiness and other motor deficits at two months of age.
“Our findings suggest that C9ORF72 mutations impair neurodevelopment in utero in patients with ALS and FTD, and that this could potentially contribute to the onset of disease symptoms later in life,” said Ichida.
Other co-authors are Alicia M. Quihuis, Shu-Ting Hung, Jonathan Chang, Nomongo Dorjsuren, Balint Der, Kim A. Staats, Yingxiao Shi, Naomi S. Sta Maria, and Russell E. Jacobs from USC.
More information:
Eric Hendricks et al, The C9ORF72 repeat expansion alters neurodevelopment, Cell Reports (2023). DOI: 10.1016/j.celrep.2023.112983
Journal information:
Cell Reports
Source: Read Full Article